Sayana® Press
Alternative Name: Depo-subQ + Uniject
Description: Sayana Press contains 104 mg of depot medroxyprogesterone acetate (DMPA) per 0.65 mL dose and is administered via subcutaneous (SC) injection using the Uniject® injection system, a small pre-filled single use device.
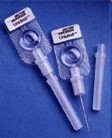
Product Details
Duration: 3 months
Regimen:
Dose: 104 mg
Active Pharmaceutical Ingredient (API):
Status Details
Developer: , ,
Status Details: - Pfizer registered depo-subQ provera 104, the same drug in Sayana Press, with the US FDA in 2004.
Sayana and Sayana Press are also registered with the UK Medicines and Healthcare Products Regulatory Agency (MHRA). It was approved in the European Union via procedure number UK/H/0960/002UK/H/0960/002.
- Sayana Press is being introduced into Bangladesh, Burkina Faso, Niger, Senegal, and Uganda in 2014.
- Pfizer registered depo-subQ provera 104, the same drug in Sayana Press, with the US FDA in 2004. Sayana and Sayana Press are also registered with the UK Medicines and Healthcare Products Regulatory Agency (MHRA). It was approved in the European Union via procedure number UK/H/0960/002UK/H/0960/002.
- Sayana Press is being introduced into Bangladesh, Burkina Faso, Niger, Senegal, and Uganda in 2014.
Additional Information
References: PATH. Frequently Asked Questions About Sayana® Press: https://www.path.org/publications/detail.php?i=2742
PATH. Frequently Asked Questions About Sayana® Press: https://www.path.org/publications/detail.php?i=2742